Z-CILI-TC
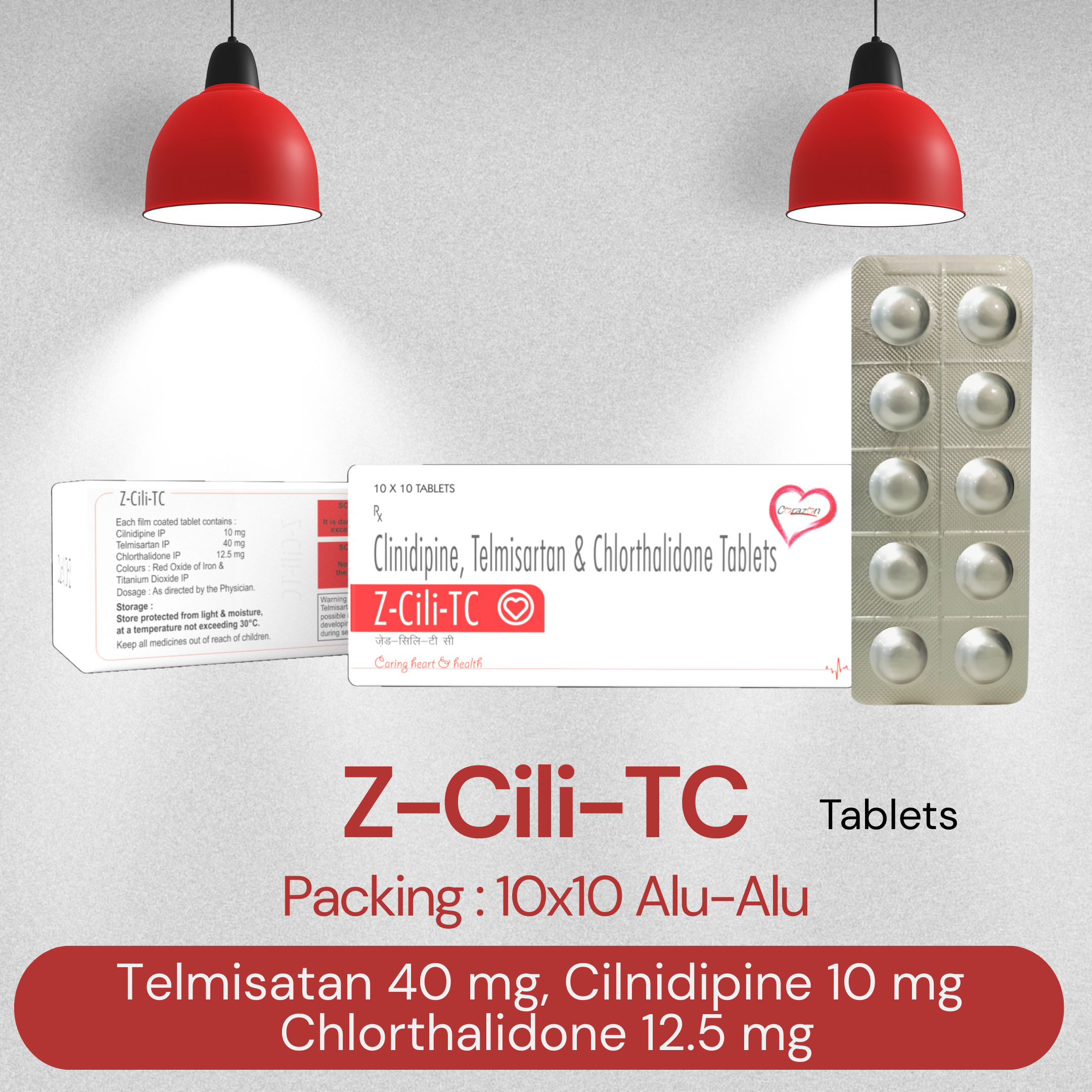
Brand Name : Telmisatan+ Cilnidipine and Chlorthalidone Tablet
Composition : Z-CILI-TC
Packing Type : 10x10
Product Price : Rs. 1190
ZCILI-TC is a triple-combination antihypertensive medication that contains Telmisartan, Cilnidipine, and Chlorthalidone, specially formulated for the effective management of high blood pressure (hypertension). This advanced combination therapy provides superior blood pressure control by targeting multiple pathways responsible for elevated blood pressure.
The composition Telmisartan + Cilnidipine + Chlorthalidone Tablet works by relaxing blood vessels, reducing fluid retention, and improving blood circulation, thereby lowering blood pressure and reducing the risk of heart attack, stroke, and kidney complication
Composition of the ZCILI-TC tablet
The ZCILI-TC tablet contains the following ingredients :
- Telmisartan
- Cilnidipine
- Chlorthalidone
- Excipients q.s.
Mechanism of action
Cilnidipine acts on the L-type calcium channels and blocks the incoming calcium and suppressing the contraction of blood vessels, thereby reducing blood pressure. It also acts on the N-type calcium channel located at the end of the sympathetic nerve, inhibiting the emission of nor-epinephrine and decreases the blood pressure.
Telmisartan interferes with the binding of angiotensin II to the angiotensin II AT1-receptor by binding reversibly and selectively to the receptors in vascular smooth muscle and the adrenal gland.
Chlorthalidone is a thiazide-like diuretic used for the treatment of hypertension and for the management of oedema caused by conditions such as heart failure or renal impairment.
Dosage and Administration
The tablets must be taken as per the prescription of the doctor. Some of the common recommendations for the tablet are :
- Usually taken once daily
- Take at the same time each day
- Can be taken with or without food
- Do not stop medication without medical advice
Pharmacology
Absorption
Cilnidipine absorbs very rapidly and attains a maximum peaked concentration after 2 hours. Its distribution tends to be higher in the liver as well as in kidneys, plasma and other tissues.
Telmisartan’s bioavailability depends on dosage. Food slightly decreases the bioavailability (a decrease of about 6% is seen when the 40-mg dose is administered with food).
Chlorthalidone prevents reabsorption of sodium and chloride through inhibition of the Na+/Cl- symporter in the cortical diluting segment of the ascending limb of the loop of Henle.
Distribution
Clinidipine’s distribution is higher in the liver as well as in kidneys, plasma and other tissues. It doesn’t present a high accumulation in the tissue after repeated oral administration. Cilnidipine is reported to present very low bioavailability, approximately 13%.
The Volume of distribution of Telmisartan is 500 L.
Chlorthalidone has been shown to rapidly concentrate within erythrocytes and subsequently equilibrate via a slow diffusion back into the serum compartment, resulting in a large volume of distribution.
Excretion
Cilnidipine excretes through the urine in a proportion of 20% of the administered dose and 80% is eliminated by the feces.
Most the Telmisartan’s administered dose (>97%) eliminates unchanged in feces via biliary excretion; only minute amounts were found in the urine.
Approximately 50% of the administered dose is excreted unmetabolized through the kidney, and excretion is characterized by biphasic elimination with a rapid phase followed by a slow secretory phase.
Precautions
Inform your physician, if you are
• Not not safe to use during pregnancy
• Precautions should be taken if you have skin allergy or breathing problems
Contraindications
• Cardiogenic shock, recent MI or acute unstable angina; severe aortic stenosis
Storage Instructions
Store in a cool and dry place
Protect from sunlight and moisture
Keep out of reach of children
Frequently Asked Question
Address
SCO 5-6, Zirakpur-Panchkula-Kalka Hwy, near Hotel Sunpark, Wadhawa Nagar, Dhakoli, Zirakpur, Punjab 140603
Contact No.
+91-7087571536
Email
arlakcorazon@gmail.com



