CORAPARIN 40MG
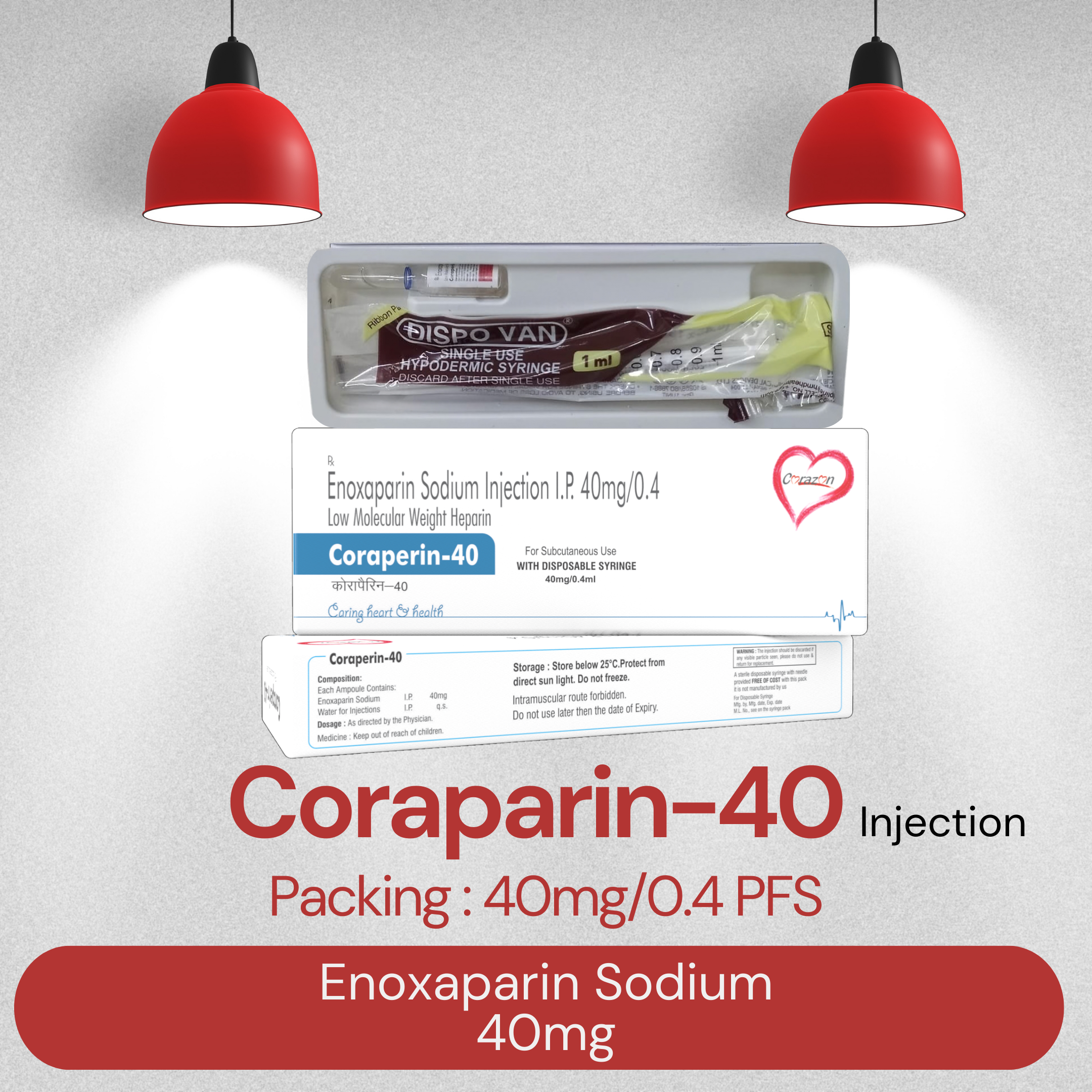
Brand Name : Enoxaparin Sodium 40mg Injection
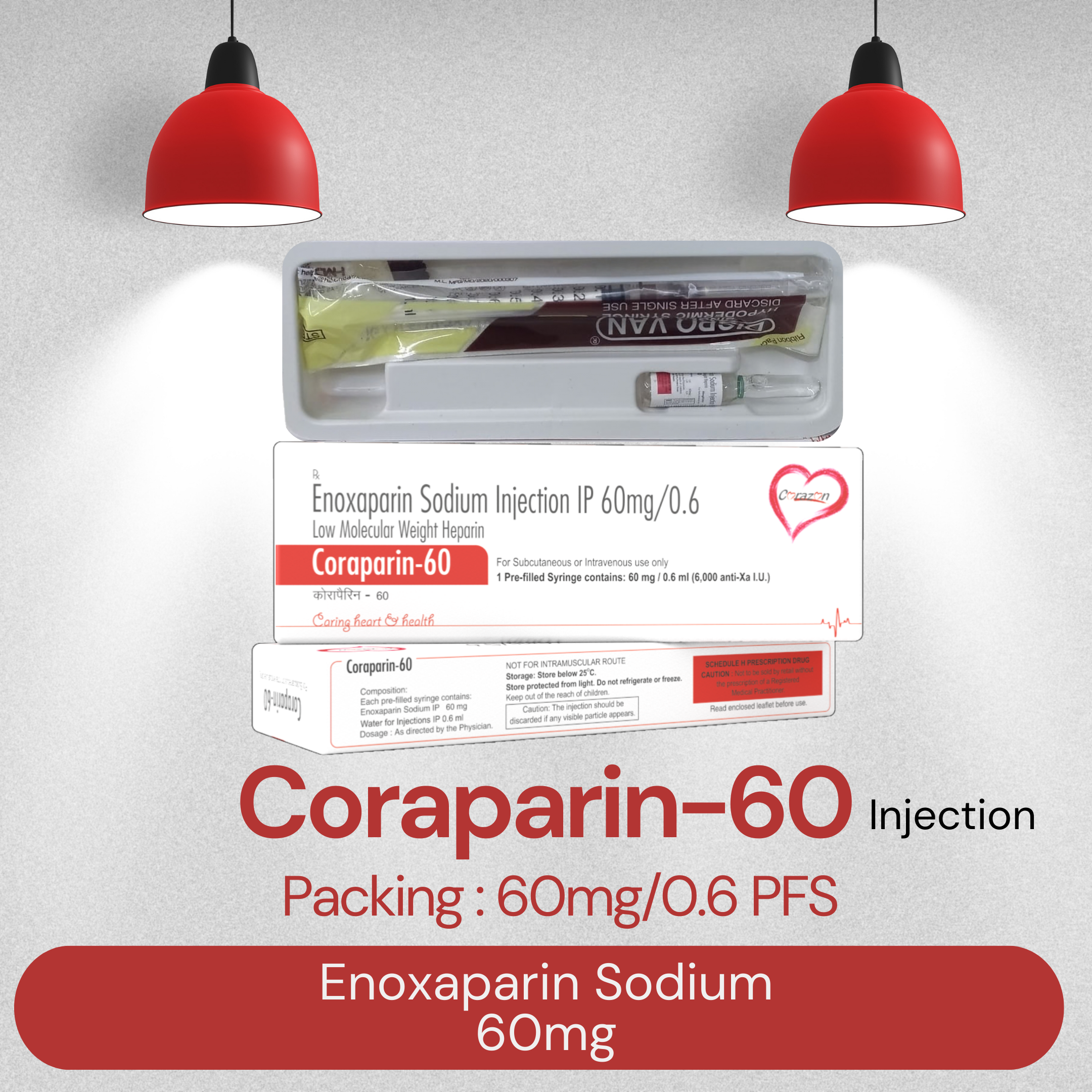
Composition : CORAPARIN 40MG
Packing Type : PFS
Product Price : Rs. 475
ENOXAPARIN SODIUM 40 MG INJECTION is related to the drug category named anticoagulant, and it is useful to treat harmful blood clots in the human body. This medication works by stopping the formation of new clots and also existing clots from getting any bigger. A blood clot is a kind of health problem in which a clump of blood forms in the vessels and blocks the regulation of blood flow. Blood clots are a natural response to injury, as they help to stop bleeding by forming a clot that plugs the damaged blood vessel. However, blood clots can also occur inside blood vessels without injury, leading to serious health complications.
Patients should not self-administer ENOXAPARIN SODIUM 40 MG INJECTION medication, because it may lead patients to harmful effects. Patients may feel some common side effects with the use of the drug, and these adverse effects include, bleeding, headache, increment of liver enzymes, skin rash, itchy red skin, bruising, or pain at the injection site. The symptoms of side effects do not need any medical attention, because these effects resolve after some time but if these effects gradually increased then the patient should take immediate consultation with professional healthcare.
Those patients should not use ENOXAPARIN SODIUM 40 MG INJECTION injections who are facing any allergic reaction or feel any interaction with any of its ingredients. Pregnant and breastfeeding ladies should use this medicine after informing their doctor. The user should tell his doctor about any health problems like stomach ulcers, high blood pressure, brain surgery, recent bleeding stroke, and stroke, and liver, kidney disorders. With the use of this drug potassium level in the blood of some patients increases which causes overweight, and underweight problems in patients. This is a prescription drug and the dose with duration is recommended by the doctor to the patients after monitoring the health condition of the patients.
Common Side Effects Of Use Of ENOXAPARIN SODIUM 40 MG INJECTION
It is not necessary that every user of ENOXAPARIN SODIUM 40 MG INJECTION injection feel the side effects with use. In other words, we can say that it varies from person to person. Here is a list of some symptoms related to common side effects.
Bleeding
Increases in liver enzymes
Bruise more easily than usual
Pink patches on your skin
Skin rash (hives, urticarial)
Itchy red skin
Pain at the site of injection
Decreased red blood cell count
High platelet counts in the blood
Headache
Drug To Drug Interaction of ENOXAPARIN SODIUM 40 MG INJECTION
ENOXAPARIN SODIUM 40 MG INJECTION injections may interact with other categories of medicines like painkillers, blood thinners, and asthma drugs. Here are some names of medications that have interactions with this medicine.
- ASPIRIN
- IBUPROFEN
- HEPARIN
- WARFARIN
- RIVAROXABAN
- DEXTRAN INJECTION
- PREDNISOLONE
- DEXAMETHASONE
Safety Advise For ENOXAPARIN SODIUM 40 MG INJECTION
The injection is a medication that contains the blood thinner substance enoxaparin. It is useful in stopping the formation of blood clots. Following is some advice for safety purposes for using the medicine.
- After instruction by the healthcare provider patients should use the injection properly.
- It is important for the user to follow instructions carefully and not deviate from the prescribed dose.
- Patients should be aware of the blood thinning signs because injection thins the blood that increases the risk of bleeding, such as bruising, bleeding gums, or blood in your urine or stool.
- In case of having a history of bleeding disorders, liver or kidney disease, or any other medical conditions. Patients should inform their doctor.
- Avoid the use of other medications that can increase the risk of bleeding, such as aspirin or no steroidal anti-inflammatory drugs (NSAIDs).
- Using a soft-bristled toothbrush can help the patient to reduce the risk of bleeding gums.
- Attend regular check-ups for managing doses of medication according to a health condition.
Address
SCO 5-6, Zirakpur-Panchkula-Kalka Hwy, near Hotel Sunpark, Wadhawa Nagar, Dhakoli, Zirakpur, Punjab 140603
Contact No.
+91-7087571536
Email
arlakcorazon@gmail.com